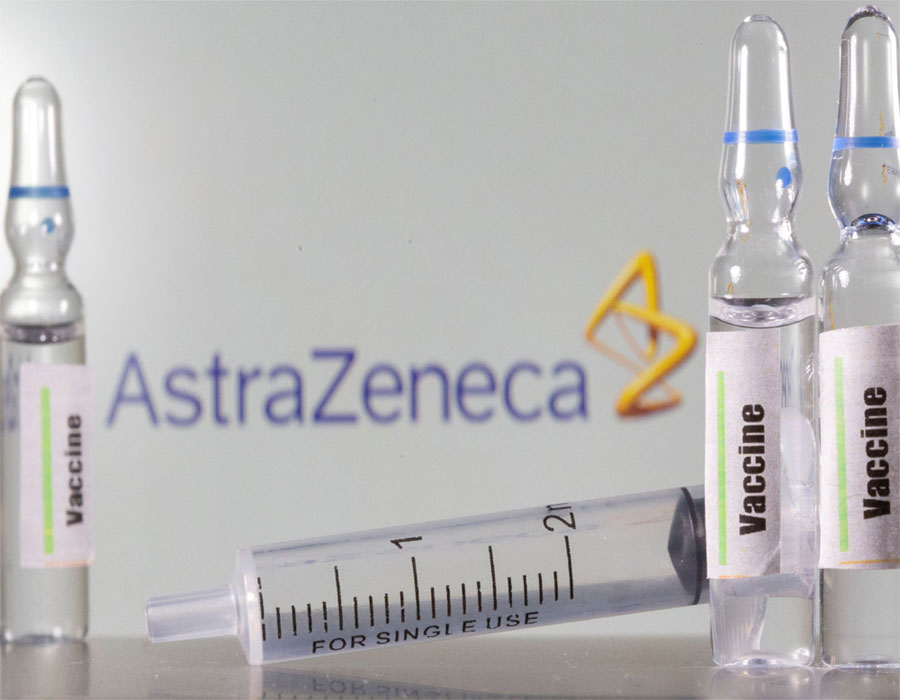
The European Commission has granted conditional marketing authorization for the Covid-19 vaccine developed by British company AstraZeneca and Oxford University, although the bloc seems unlikely to get the amount of doses as agreed at the moment.
"Securing safe vaccines for Europeans is our utmost priority. With the AstraZeneca vaccine now authorized, 400 million additional doses will be available in Europe," European Commission President Ursula von der Leyen said on Friday.
Von der Leyen said that she expected AstraZeneca to deliver the doses as agreed so that Europeans can be vaccinated as soon as possible, reports Xinhua news agency.
"We will continue doing all we can to secure more vaccines for Europeans, our neighbors and our partners worldwide."
The European Union has been squabbling with the British company over the latter's announcement that it may deliver considerably fewer doses than promised for the first quarter.
The bloc published the contract with AstraZeneca on Friday, and meanwhile launched a transparency measure to regulate vaccines export from the union.
This is the third Covid-19 vaccine authorized in the EU, after Pfizer/BioNTech and Moderna vaccines were given the greenlights in late December and early January.
The authorization on Friday was granted hours after the European Medicines Agency (EMA) issued a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and quality of the vaccine.
Combined results from four clinical trials in the UK, Brazil and South Africa, involving around 24,000 people, showed that the vaccine was safe and effective in preventing Covid-19 in people from 18 years of age, according to the EMA.
Based on data relating to patients who received standard doses followed by second standard doses, the conclusion was drawn that the vaccine demonstrated nearly 60 percent efficacy in the clinical trials, it added.
(Courtesy: IANS)







 OpinionExpress.In
OpinionExpress.In












Comments (0)